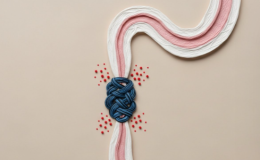
The human body has two major barriers -the gut barrier and the blood-brain barrier (BBB)- in addition to the placental barrier in pregnant females. The gut barrier regulates the flow of nutrients and signals molecules in the body and prevents the entry of microorganisms,food residue,and harmful substances. The gut microbiota regulates the development and function of these barriers and many factors like stress,alcohol use, unhealthy diet damage the gut barrier, increase intestinal permeability and allow microorganisms to pass through to the body that could not do so before;this syndrome is called leaky gut.
The altered host-microbe relationship can be defined as dysbiosis when the microbial chift has pathophysiological consequences. Pathobionts are members of the symbiotic community that expand as a result of the imbalance and exert pathogenic effects on the host
In the gut, the alimentary canal is lined by epithelial cells that form the mucosa and, with few exceptions, the gastrointestinal epithelium is tied contiguously through tight junctions, where diversity among epithelial cells affect specific barrier functions. Gastric mucus and mucin, part of the extrinsic barrier consisting of secretions, also affect the barrier by slowing diffusion rates of hydrophilic molecules and aqueous solutions like gastric acid.
The gastrointestinal tract is the interface between the host and environment, and especially the small intestine has a huge surface area to control the important function of digestion and absorption of nutrients. Since the intestine is the site with the highest bacterial concentration, the host was forced to develop a strategy of tolerance to beneficial and harmless microorganisms, but also and efficient defense mechanism against pathogens and bacterial overgrowth
Disease-predisposing microbiota was found in a wide spectrum of chronic disorders. These findings have led to the speculation of a common factor in multigenic disease development. A common ground hypothesis was proposed to indicated the key roles of microbiota dysbiosis associated with a leaky gut in the pathogenesis of chronic polygenic diseases. The hypothesis, which still needs to be rigorously examined, first suggests that endogenous and exogenous factors which cause gut barrier impairment and low grade immune activation could impose selective pressure on the intestinal microbiota. The subclinical mucosal abnormalities that developed in individuals with genetic predisposition then favor the growth of opportunistic microbes with virulence emergence.
The expanded concept of a leaky gut suggest that with age, and under specific conditions, small molecule bacterial metabolic components can translocate or diffuse systemically, and even disseminate to distal sites from the gut epithelial barrier to even pass the blood brain barrier and reach the blood-cerebrospinal fluid (CSF) or other brain components
Common ground hypothesis: the initial epithelial barrier dysfunction manifested by transcellular hyperpermeability and passive bacterial internalization may instigate a selection pressure on microbiota (such as positive inforcement by anchorage and growth advantage, and negative impediment by aerotolerance and immune evasion), leading to the emergence of invasive virulent pathobionts. The selection pressure and mucosal pathobionts may cause a shift in the fecal microbial community. The bacterial internalization may also cause epithelial cytoskeletal disorganization and paracellular destruction. The combination of broken epithelial barrier and invasive pathobions results in a massive amount of bacterial translocation, which leads to clinical features of morphological damage and chronic inflammation. Mucosa-associated pathobionts have been assumed to derived from unharmful gut commensals,yet without direct evidence. One of the proposed triggers for emergence of pathogenic commensals was the need for adaptation to oxidative stress.
Mucus barrier
The intestine is lined by mucus, with an inner dense layer and an outer loosely adhering layer, which enlarges along the gastrointestinal tract and it is thickest in the colon. Especially the inner layer of the mucus represents a highly efficient first defense mechanism. Because of its high density,it prevents most bacteria from penetration and thus isolates the epithelium from the huges amounts of luminal microbiota.
Mucins are rich in glycosylation and mores than 100 different mono-,di-, or trisialylated oligosaccharides are described in human mucus. The mucins show a high conservation between individuals and therefor point to an important role in the selection of commensal intestinal bacteria (only few microorganisms are able to adhere to mucus)
Although the mucus and unstirred layers prevent some organisms and large molecules,eg, food particles, from directly accessing the epithelium, they do little to prevent flux of small molecules,ions,and water. Nevertheless, studies with experimental animals have shown that disruption of mucus production can lead to intestinal damage and inflammatory disease
Intestinal Epithelial Barrier
The intestinal epithelium separates the gut lumen from the lamina propia, and it is mainly composed of absorptive enterocytes,globet-,Paneth-, and endoenterocrine cells. The cell layer forms a physical barrier that allows the restrited paracellular transport of molecules. The tight junction are composed of several transmembrane and cytosolic proteins and the exact regulation of tight junction proteins is mandatory for the epithelial integrity.
Epithelial cells and dendritic cells recognize bacteria via so called pattern recognition receptors (PPR) such as Toll-like or Nod-like receptors. Toll-like receptor 4 (TLR4) is mainly triggered by lipopolysaccharide of Gram-negative bacteria, whereas TLR2 is activated by peptidoglycan and lipoteichoic acids, the main cell wall components of Gram-positive bacteria.